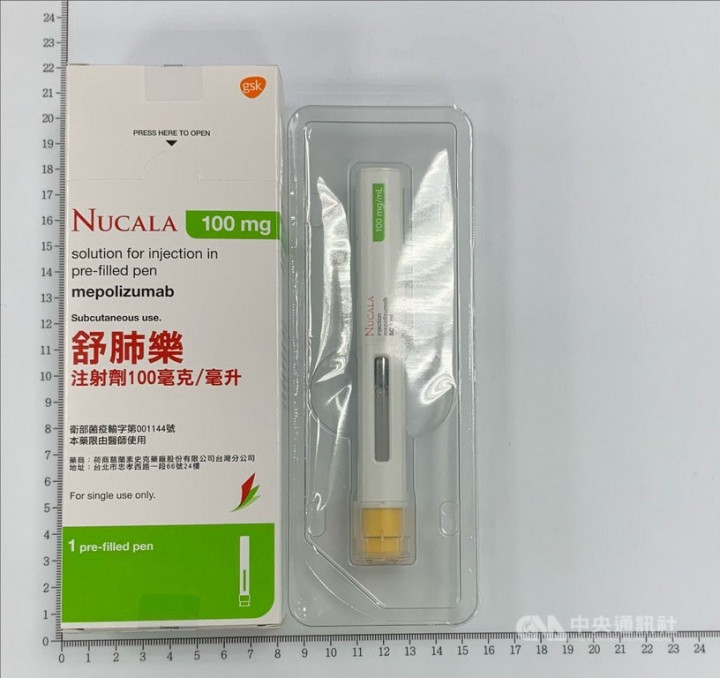
Taipei, July 19 (CNA) Taiwan's Food and Drug Administration (FDA) said Monday that as a precautionary measure, it has ordered the recall of a batch of Nucala Solution for Injection, a prescription medicine used to treat severe asthma, because of a potential quality defect.
The recall is set to be completed by Aug. 8, said Hung Kuo-teng, head of the FDA's Medicinal Products Division, at a joint press interview.
He said the Nucala Solution for Injection in Pre-filled Pen 100mg/mL is a new drug that was first sold in Taiwan at the end of last year. Currently, only one of the products has been sold locally, he added.
The drug is produced by the British pharmaceutical company GlaxoSmithKline as a supporting therapy for patients aged 12 or older with severe refractory eosinophilic asthma whose condition is poorly controlled.
Eosinophilic asthma is a type of severe asthma marked by high levels of white blood cells, called eosinophils.
However, in foreign markets, a fiber has been found in the syringe wall of the product, which has been identified as a quality defect, Hung said.
In addition, the products Taiwan imported under lot number FA9M were made with semi-finished items from the same batch number of materials used to make the defective products.
"For the sake of patient medication safety," the FDA initiated the recall as a precaution, Hung said.
According to FDA technical specialist Pan Hsiang-ying, lot number FA9M contained 545 boxes of Nucala Solution for Injection 100mg/mL, each with one injector.